U.S. Healthcare Passive Cold Chain Packaging Market Summary
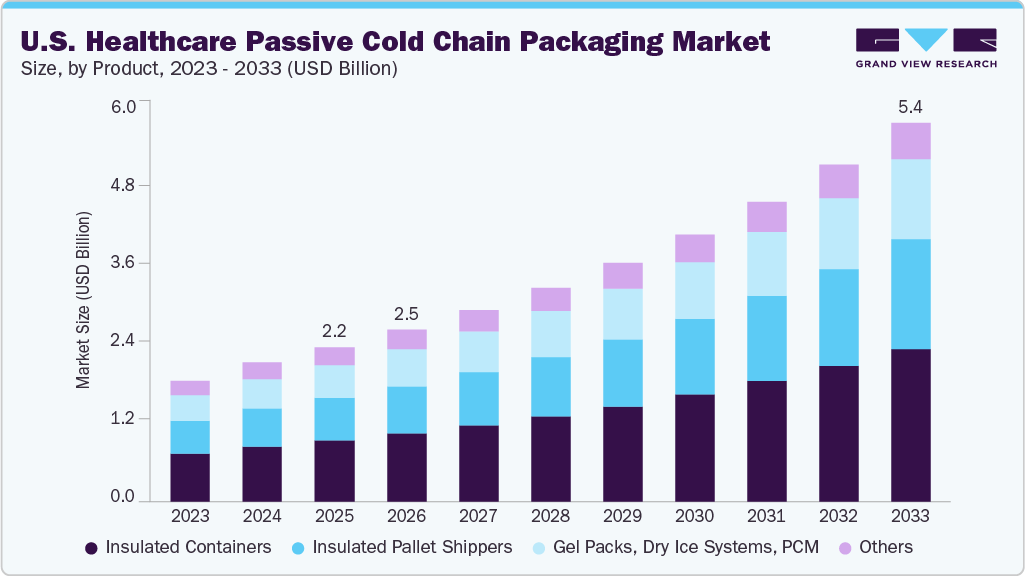
The U.S. healthcare passive cold chain packaging market size was valued at USD 2.22 billion in 2025 and is projected to reach USD 5.45 billion by 2033, expanding at a CAGR of 11.9% from 2026 to 2033. The industry is primarily driven by the rising production and distribution of temperature-sensitive biologics, vaccines, and advanced therapies, which demand reliable, validated passive temperature control solutions.
Key Market Trends & Insights
- By product, the insulated pallet shippers segment is expected to grow at a considerable CAGR of 12.7% from 2026 to 2033 in terms of revenue.
- By temperature requirement, the frozen (-20 °C to ±5 °C) segment is expected to grow at a considerable CAGR of 12.6% from 2026 to 2033 in terms of revenue.
- By end use, the pharmaceutical & biopharmaceutical companies segment is expected to grow at a considerable CAGR of 12.8% from 2026 to 2033 in terms of revenue.
Market Size & Forecast
- 2025 Market Size: USD 2.22 Billion
- 2033 Projected Market Size: USD 5.45 Billion
- CAGR (2026-2033): 11.9%
Additionally, cost-efficiency and operational simplicity of passive systems make them widely adopted across domestic healthcare logistics, especially for last-mile and direct-to-patient shipments.
The rising demand for temperature-sensitive biologics and vaccines is one of the most significant growth drivers for the U.S. healthcare passive cold chain packaging industry. Unlike conventional pharmaceuticals, biologics such as monoclonal antibodies, cell and gene therapies, and recombinant proteins are highly vulnerable to temperature variations. Even limited exposure outside recommended ranges can compromise product efficacy, making reliable passive cold chain solutions essential for protecting drug quality during storage and distribution.

The U.S. remains a global leader in biologics development and commercialization, supported by strong pharmaceutical innovation and increasing approvals of advanced therapies. These products require distribution across hospitals, specialty pharmacies, clinical trial sites, and increasingly direct-to-patient channels. Passive cold chain packaging, including insulated shippers, phase change materials, and gel packs, offers a scalable and cost-effective approach to ensure temperature control without the operational burden of fully active refrigeration systems.
Vaccines further strengthen this demand, particularly with the expansion of immunization programs, seasonal influenza supply chains, and broader preparedness for public health emergencies. Vaccine distribution involves high shipment volumes, variable transit durations, and complex last-mile delivery requirements. Passive packaging is widely preferred due to its flexibility, lower cost structure, and validated performance across diverse climate and transportation conditions.
Moreover, the increasing adoption of e-commerce in healthcare logistics is becoming a major growth driver for the industry. Healthcare distribution is expanding beyond traditional hospital and wholesaler channels toward digital ordering platforms, specialty pharmacy networks, and direct-to-patient delivery models. For example, specialty pharmacies shipping biologics for rheumatoid arthritis or multiple sclerosis now rely heavily on insulated parcel shippers to ensure products remain within the required 2-8°C range during doorstep delivery. This structural shift is significantly increasing demand for validated passive cold chain packaging solutions.
Market Concentration & Characteristics
Stringent regulatory requirements in the United States are another major growth factor. Agencies like the U.S. Food and Drug Administration (FDA) and CDC enforce rigorous Good Distribution Practices (GDP) and validation standards for cold chain packaging to protect product quality and patient safety. Healthcare and biopharmaceutical companies must demonstrate that their packaging and logistics meet these standards, which drives demand for pre-qualified passive systems with proven thermal performance, traceability, and documentation. This regulatory environment reduces reliance on untested or low-cost packaging and accelerates adoption of validated, high-performance solutions.

The growth of direct-to-patient delivery, clinical trials, and decentralized healthcare models is increasing reliance on passive cold chain packaging. As more therapies and vaccines are shipped directly from manufacturers to clinics, pharmacies, or patients’ homes, smaller, reliable passive systems are needed for last-mile and short-haul distribution. These solutions offer operational simplicity, scalability, and lower total cost compared with powered active systems, making them well-suited for a wide range of domestic healthcare logistics scenarios. Furthermore, this trend is amplified by the expansion of e-commerce in healthcare, where medications and biologics are increasingly ordered and shipped through digital channels.
Product Insights
The insulated containers segment dominated the U.S. industry in 2025, accounting for over 39.0% of revenue, primarily due to their superior thermal protection, durability, and ability to maintain stable temperatures for biologics, vaccines, and specialty drugs during transit. These containers are widely adopted by pharmaceutical and biopharmaceutical companies because they offer validated performance for refrigerated and frozen shipments across long distribution routes.
The insulated pallet shippers segment is projected to expand at the fastest CAGR of 12.7% during the forecast period, driven by the growing need for bulk transportation of high-value biologics, vaccines, and temperature-sensitive pharmaceuticals across long-distance supply chains. These shippers provide extended thermal stability, high payload capacity, and cost efficiency for large-volume distribution, making them increasingly preferred by pharma manufacturers and logistics providers. Additionally, the rapid expansion of clinical trial shipments and large-scale immunization programs is accelerating the adoption of pallet-level passive cold chain solutions.
Temperature Requirement Insights
The refrigerated/chilled (2°C to 8°C) segment dominated the U.S. healthcare passive cold chain packaging market in 2025, accounting for over 46.0% revenue share, due to the high volume of biologics, vaccines, insulin, and specialty injectable drugs requiring this standard temperature range. This category represents the most widely used cold chain condition across pharmaceutical distribution, hospital supply networks, and retail pharmacy logistics. Additionally, strict FDA compliance requirements and the expansion of routine immunization and chronic disease treatments further strengthened demand for reliable passive chilled packaging solutions.
The frozen (-20°C to ±5°C) segment is expected to grow at the fastest CAGR of 12.6% during the forecast period, driven by the rising distribution of temperature-sensitive biologics, advanced vaccines, and specialty pharmaceuticals that require frozen storage for stability. Increasing clinical trial activity, expansion of gene-based therapies, and greater demand for long-distance shipping of high-value drugs are accelerating the adoption of passive frozen packaging systems. Additionally, improvements in phase-change materials and high-performance insulation are enhancing the reliability of frozen cold chain solutions, supporting rapid market growth.
End Use Insights
The pharmaceutical & biopharmaceutical companies segment dominated the U.S. healthcare passive cold chain packaging market in 2025, capturing over 40.0% market share, as these firms are the primary producers and distributors of temperature-sensitive biologics, vaccines, and specialty injectable drugs. The segment is also expected to grow at the fastest CAGR of 12.8%, driven by the rapid expansion of biologics pipelines, cell and gene therapies, and increasing clinical trial shipments requiring validated passive packaging. Additionally, strict regulatory compliance and the shift toward direct-to-patient and decentralized distribution models are further accelerating demand from pharma and biopharma manufacturers.
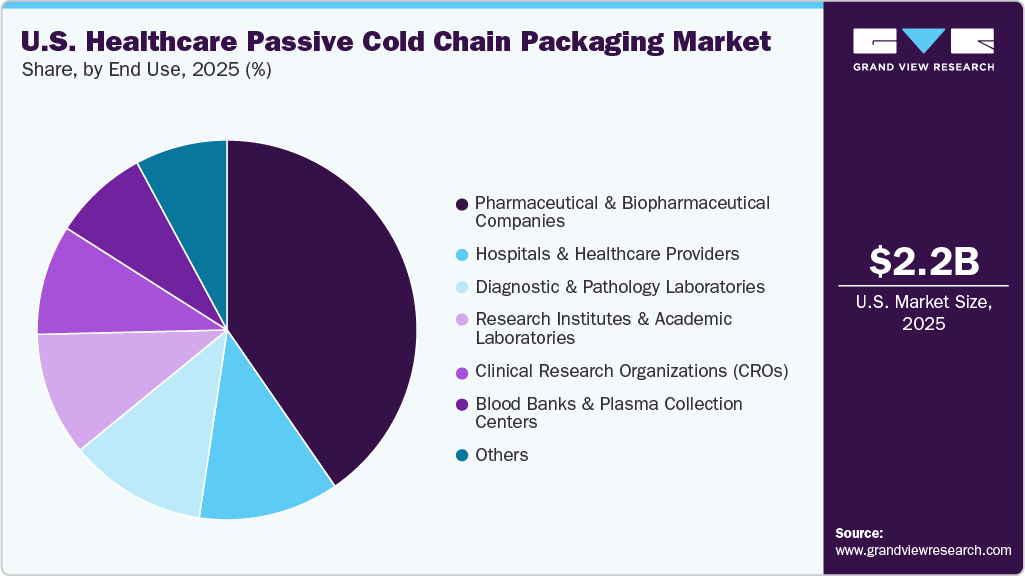
The hospitals & healthcare providers segment represents a significant growth area in the U.S. healthcare passive cold chain packaging market, driven by the increasing administration of temperature-sensitive vaccines, biologics, and specialty drugs within clinical settings. Hospitals require reliable passive packaging solutions to ensure safe storage and internal distribution of critical therapies, particularly for oncology, immunology, and emergency care treatments. Additionally, the rise in outpatient services, hospital-based specialty pharmacies, and stricter compliance with cold chain handling standards is strengthening demand from this end-use segment.
Key U.S. Healthcare Passive Cold Chain Packaging Company Insights
The competitive environment of the U.S. healthcare passive cold chain packaging market is characterized by intense, innovation-driven competition among a moderate number of established players that focus on performance, regulatory compliance, and service capabilities rather than on price alone. Major players such as ThermoSafe, Cold Chain Technologies, Peli BioThermal LLC, Cryopak, CSafe, Intelsius, and TemperPack dominate the landscape with validated passive solutions tailored for biologics, vaccines, and specialty pharmaceuticals; their deep technical expertise, extensive testing, and long-standing relationships with large pharmaceutical and biopharmaceutical customers provide a strong competitive advantage. competition increasingly revolves around product performance, duration of temperature control, digital integration, and supply chain support services, with companies investing in capacity expansion, service centers, and sustainable innovations to maintain or grow market share.
Key U.S. Healthcare Passive Cold Chain Packaging Companies:
- ThermoSafe
- Cold Chain Technologies
- Peli BioThermal LLC
- Cryopak
- CSafe
- Intelsius
- TemperPack
- Coldkeepers, LLC
- Polar Tech Industries
- Creative Packaging Co.
Recent Developments
-
In November 2025, Sonoco Products Company completed the sale of its ThermoSafe temperature-assured packaging business to private equity firm Arsenal Capital Partners. The divestiture refocuses Sonoco on core metal and fiber packaging, while ThermoSafe will operate under new ownership to accelerate investment in specialized pharma shippers and service capabilities relevant to U.S. life sciences customers.
-
In March 2025, Peli BioThermal LLC introduced Vēro One, a curbside-recyclable dry-ice shipper designed for deep-frozen shipments. The single-use, recyclable design targets situations where reusable systems are impractical, providing U.S. shippers with a lower-waste option for ultra-cold clinical and small commercial lanes.
U.S. Healthcare Passive Cold Chain Packaging Market Report Scope
|
Report Attribute
|
Details
|
|
Market size value in 2026
|
USD 2.47 billion
|
|
Revenue forecast in 2033
|
USD 5.45 billion
|
|
Growth rate
|
CAGR of 11.9% from 2026 to 2033
|
|
Historical data
|
2021 – 2024
|
|
Forecast period
|
2026 – 2033
|
|
Quantitative units
|
Revenue in USD million/billion, and CAGR from 2026 to 2033
|
|
Report coverage
|
Revenue forecast, competitive landscape, growth factors, and trends
|
|
Segments covered
|
Product, temperature requirement, end use, region
|
|
Country scope
|
U.S.
|
|
Key companies profiled
|
ThermoSafe; Cold Chain Technologies; Peli BioThermal LLC; Cryopak; CSafe; Intelsius; TemperPack; Coldkeepers, LLC; Polar Tech Industries; Creative Packaging Co.
|
|
Customization scope
|
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional, and segment scope.
|
|
Pricing and purchase options
|
Avail customized purchase options to meet your exact research needs. Explore purchase options
|
U.S. Healthcare Passive Cold Chain Packaging Market Report Segmentation
This report forecasts revenue growth at the country level and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the U.S. healthcare passive cold chain packaging market report based on product, temperature requirement, end use, and region:
-
Product Outlook (Revenue, USD Million, 2021 – 2033)
-
Insulated Containers
-
Insulated Pallet Shippers
-
Gel Packs, Dry Ice Systems, PCM
-
Others
-
-
Temperature Requirement Outlook (Revenue, USD Million, 2021 – 2033)
-
Refrigerated / Chilled (2°C to 8°C)
-
Frozen (−20 °C to ±5 °C)
-
Cool / Controlled Chilled (8 °C to 15 °C)
-
Controlled Room Temperature (CRT)
-
Deep Frozen (−40 °C to −60 °C)
-
-
End Use Outlook (Revenue, USD Million, 2021 – 2033)
-
Pharmaceutical & Biopharmaceutical Companies
-
Hospitals & Healthcare Providers
-
Diagnostic & Pathology Laboratories
-
Research Institutes & Academic Laboratories
-
Clinical Research Organizations (CROs)
-
Blood Banks & Plasma Collection Centers
-
Others
-
Frequently Asked Questions About This Report
b. The key players in the U.S. healthcare passive cold chain packaging market include ThermoSafe; Cold Chain Technologies; Peli BioThermal LLC; Cryopak; CSafe; Intelsius; TemperPack; Coldkeepers, LLC; Polar Tech Industries; and Creative Packaging Co.
b. The U.S. healthcare passive cold chain packaging market was estimated at around USD 2.22 billion in the year 2025 and is expected to reach around USD 2.47 billion in 2026.
b. The U.S. healthcare passive cold chain packaging market is expected to grow at a compound annual growth rate of 11.9% from 2026 to 2033 to reach around USD 5.45 billion by 2033.
b. The pharmaceutical & biopharmaceutical companies segment dominated the U.S. healthcare passive cold chain packaging market due to the rapid expansion of biologics pipelines, cell and gene therapies, and increasing clinical trial shipments requiring validated passive packaging.
b. The market is primarily driven by the rapid rise in temperature-sensitive biologics, vaccines, cell and gene therapies, and specialty drugs that require reliable thermal protection, alongside stringent regulatory requirements and expanding biopharmaceutical production and distribution activities.
link





More Stories
CHAI’s AI oversight ambitions falter with scrapped AI labs
Nutrition in healthcare brings opportunity for industry growth and innovation
Lewis and Clark County job industry sees biggest growth in healthcare