Materials
3,4-Ethylenedioxythiophene (EDOT, 99%) was purchased from Thermo Fisher Scientific, while iron (III) p-toluenesulfonate hexahydrate (FeTos) was obtained from Sigma-Aldrich. The polysaccharide Sacran was extracted from Suizenji Nori cyanobacteria by Maiko Okajima and Tatsuo Kaneko in Japan and transferred to Austria. The solution additives 4-dodecylbenezenesulfonic acid (DBSA), glycerol and (3-glycidyloxypropyl) trimethoxysilan (GOPS) were acquired from Fluka, Carl Roth and Grolman, respectively. Commercial poly(3,4-ethylenedioxythiophene): poly(styrenesulfonate) (PEDOT:PSS, CleviosTM PH 1000) was obtained from Heraeus Electronics GmbH (Hanau, Germany). Prior to usage, the PEDOT:PSS dispersion was filtered through a polyvinylidene fluoride filter with a pore size of 0.45 µm. All other reagents were used as received without further purification. The proteinase K from Tritirachium album was purchased from Thermo Fischer Scientific.
Purified 18 MΩ ultrapure water was obtained by the Arium® Mini laboratory water system.
Extraction of Sacran
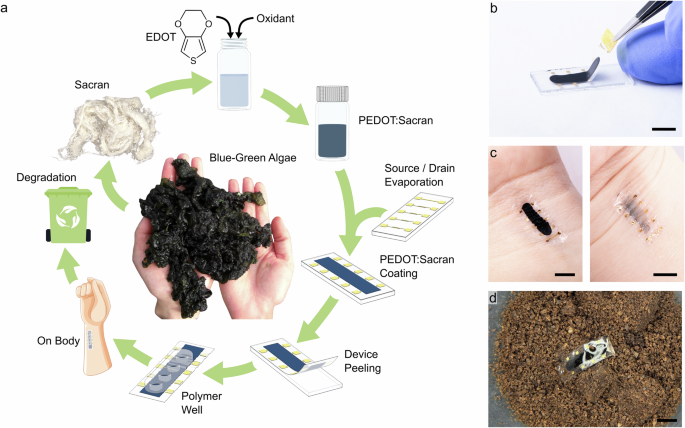
Sacran was extracted from Aphanothece sacrum (A. sacrum) by the following procedure31,32,68. The crude A. sacrum biomaterials were freeze-thawed and washed in distilled water at least three times to remove any water-soluble matter. The biomaterials were further washed five times using a large amount of isopropanol with shaking to remove hydrophobic pigments such as chlorophylls. The isopropanol-washed samples were then put into a 0.1 M NaOH and heated to around 100°C and agitated for 4 h to yield the transparent solution. The solution was neutralized with HCl until the pH decreased to 8.0–9.0 and then was filtered. The filtrate was concentrated by a rotary evaporator to create a highly viscous solution. The viscous solution was slowly poured into 60% isopropanol to precipitate a white fibrous material. The extracted yield of Sacran was very high ca. 70 wt % in dried materials of A. sacrum.
The absolute molecular weight, Mw, was measured by multi-angle static light scattering (MALLS; detection angle from 15° to 40°) and reported previously43. From Zimm-Berry plot with an error of 1.4%, the absolute Mw, radius of gyration, Rg, and second virial coefficient, A2, for sacran were estimated at 2.35 × 107 g mol−1, 402 nm, and 4.53 × 10−4 mole cm3 g−2, respectively. Sacran was mainly composed of various monosaccharides such as Glc, Gal, Man, Xyl, Rha, and Fuc with a composition of 25.9, 11.0, 10.0, 16.2, 10.2, and 6.9, as a result of GC analyses32. Sacran was sulfated in 10 mol% (CHN S elemental analyses) and had a carboxylic acid group in 22 mol% (carbazole-sulfuric acid method (525 nm)) to monosaccharide residues32.
Synthesis of PEDOT:Sacran
The synthesis, applied for the PEDOT:Sacran dispersion, follows the procedure described by Matura et al.29. First, a Sacran dispersion in a total concentration of 1 mg mL−1 was prepared in 18 MΩ water, whereby the dispersion was vigorously stirred at room temperature for 2 h as well as sonicated in an ultrasonic bath. Next, EDOT monomer was injected in the different respective weight-to-weight (w/w) ratios of monomer to Sacran (1:1, 2.5:1, 5:1, and 7.5:1) followed by 2 h of additional stirring. Subsequently, 2 molar equiv. of iron (III) p-toluene sulfonate hexahydrate (with respect to EDOT) was added into the mixture. The dispersion was heated to 60 °C for 20 h under continuous stirring. After completion of the oxidative chemical polymerization indicated by the dark blue color of the solution, the dispersion was diluted with MΩ water, centrifuged at a speed of 5500 rpm for 3 min and re-dispersed in fresh MΩ water. This process was repeated four times until a neutral pH was reached. Then, the dispersion was sonicated for 30 min followed by a final consecutive centrifugation and re-dispersing step. Next, the dispersion was dialyzed for 24 h by changing the water periodically. Thereby, a dialysis membrane with a molecular weight cut-off (MWCO) of 12–14 kDa was used. The purified biocomposite dispersion was then directly used for the preparation of the OECT devise in an approx. concentration of 10 mg mL−1.
To test the redispersability, the dispersed solution of the PEDOT:Sacran was dried by vaporizing the water, yielding a dried solid biocomposite powder. For the preparation of sample dispersion with a concentration of 10 mg mL−1 the solid PEDOT-Sacran powder was re-dispersed in a respective amount of MΩ water and sonicated with a UP50H high-power processor (Hielscher Ultrasound Technology) at 60 W with a 0.6 s pulse for 10 min.
Synthesis of PEDOT:Tosylate was performed by following the same preparation procedure as for PEDOT:Sacran. The oxidative chemical polymerization was also performed using only EDOT with iron (III) p-toluene sulfonate as the oxidant in 2 molar equiv. Here, no Sacran was included. The tosylate (Tos) anion from the oxidant served as a doping counterion and stabilized the oxidized cationic PEDOT.
Material characterization
Attenuated total reflectance–Fourier-transform infrared (ATR-FTIR) spectra were recorded utilizing a Bruker Vertex 80 FTIR spectrometer equipped with a Bruker Platinum diamond attenuated total reflectance (ATR) unit. The materials and dispersion were measured after drying.
X-ray photoelectron spectroscopy (XPS) measurements were performed on a Theta Probe XPS-system (Thermo Fisher, GBR), which features a monochromated Al-Kα X-ray source with an energy of 1486.6 eV. The spot size on the sample surface was 400 μm in diameter. The hemispherical analyzer was set to a pass energy of 20 eV for the recorded high-resolution (HR) scans at an energy step size of 0.05 eV. The data acquisition and evaluation, as well as the control of the device, are performed via a software package (Avantage) provided by the system manufacturer. For the XPS characterization, the original stock dispersions (PEDOT:Sacran and PEDOT:PSS) were used without further additives, whereby the dispersions were drop-cast on glass slide.
Zeta potential and particle size measurements were conducted using a Malvern Zetasizer Nano ZSP (Malvern Instruments Ltd., Worcestershire, UK). The dynamic light scattering (DLS) method and electrophoretic light scattering (ELS) method were utilized for determining both particle size distribution and zeta potential. The DLS measurements via intensity were performed at 25 °C, placing 1.1 mL of the sample into a disposable cuvette (DTS0012) for the particle size, and 1 mL of sample into folded capillary cells (DTS1070) for the zeta potential determination. To prepare the sample dispersions, 10 µL of the original stock dispersion was diluted with 2 mL of MΩ water and then sonicated for 10 min using an ultrasonic processor set to 60 W with a 0.6-s pulse. Afterwards, the sample dispersions were further diluted transferring 1 mL of the prepared sample into 2 mL of MΩ water. This final sample was sonicated again under the same conditions and then at least 5 measurements were recorded for each type of measurement. The reference PEDOT:PSS dispersions were filtered through a 0.45 µm PTFE syringe filter after the dilution and sonication steps.
The zeta potential results were evaluated through 6 measurements (PEDOT:PSS), 16 (Sacran), 13 (PEDOT:Sacran, 1:1), 12 (PEDOT:Sacran 2.5:1), 24 (PEDOT:Sacran, 5:1), 24 (PEDOT:Sacran, 7.5:1) and 6 (PEDOT:Tos.), respectively. Higher particle size values (above 1000 nm) were dismissed due to possible aggregation effects leading to miscalculations. The zeta potential of 0.59 mV is recorded for the dispersant, MΩ water as control.
Thermal gravimetric analysis (TGA) data was recorded using a TGA 4000 Thermogravimetric Analyzer (Perkin Elmer) with a temperature program of heating from 30 °C to 900 °C with 10 °C min−1. The 1.5–3.8 mg sample was loaded into the cleaned crucible and then inserted into the instrument. For the TGA measurements, the PEDOT:Sacran with different wt. ratios as well as the reference materials, PEDOT:Tos and Sacran, were tested separately.
Thin-film characterization
For the characterization of the materials, we used glass substrates (1.5 cm × 1.5 cm) and ITO-coated glass (1.27 cm × 1.27 cm), which were cleaned in an ultrasonic bath with a 2% (v/v) aqueous solution of Hellmanex III, deionized water, acetone and isopropanol at 50 °C for 20 min each. Before further usage, the substrates were blow-dried with N2 gas and then treated with oxygen plasma at 100 W for 5 min in a Plasma Etch PE-24 plasma cleaner. For the absorption spectroscopy and conductivity samples of composites, a mixture of 94.5% (v/v) dispersion (PEDOT:Sacran in different weight ratios or PEDOT:PSS), 0.5% (v/v) DBSA, and 5% (v/v) glycerol was prepared, whereby subsequently 1% (v/v) GOPS was added into the dispersion. The composite dispersions were then deposited via drop-casting and via spin-coating methods on glass substrates for conductivity and absorbance measurements, respectively.
The conductivity of the films was measured using an Ossila four-point probe station with supplied software for evaluation.
Absorption spectroscopy was performed with a Lambda 1050 UV/Vis/NIR spectrometer (Perkin Elmer) with a 3D WB detector module (Perkin Elmer).
Contact angle tests were performed to evaluate the surface hydrophobicity of all sample biocomposites. Multiple 18 MΩ water droplets were measured at room temperature using an Ossila Contact Angle Goniometer and the average values were determined.
Scanning electron microscopy (SEM) analyses were conducted using the Thermo Scientific Scios 2 DualBeam instrument with an accelerating voltage of 5 kV. The PEDOT:Sacran dispersions (without additives) were deposited via spin-coating on clean ITO-coated glass substrates for the SEM measurements. The micrographs were taken at ×100,000 magnification. Prior to analysis, the samples were sputtered with gold (Au).
OECT device fabrication and characterization
The source-drain electrode pattern (channel length: L = 60 µm, channel width W = 2 mm) was prepared by fixing cleaned glass substrates onto a shadow mask (thickness = 200 µm) to reduce the shadowing effect during the metal deposition process. 10 nm chromium (Cr) and 100 nm gold (Au) were deposited sequentially by thermal evaporation at a pressure of 1–5 × 10−7 mbar. Here, Cr was utilized to improve the adhesion of Au to the substrate. Afterwards, the device channel was prepared by depositing a thin-film of the respective sample dispersion (PEDOT:Sacran or PEDOT:PSS) with additives, as mentioned above, via spin-coating using 900 rpm for 40 s, followed by 1200 rpm for 10 s. The area around the channel, as well as the contacts, were carefully wiped by using water-wetted cotton swabs before annealing the substrates at 120 °C for 30 min. In order to increase the film thickness, the spin-coating procedure was repeated whereby additionally a short annealing step of 2 min at 120 °C was employed instead of the final annealing step at 120 °C for 30 min. To deposit a thicker channel layer (approx. 10 μm thick), we drop-cast dispersions and then dried for 30 min at 100 °C followed by a final annealing step of 120 °C for 20 min.
To complete the devices, a laser-cut polymer well of 3MTM VHBTM Tape (acrylic adhesive with a conformable acrylic foam core) was fixed on the substrates to secure the aqueous electrolyte. For the device characterization, 20 µL of phosphate buffered saline (PBS) solution (50 mM, pH 7.4) was used, and a silver/silver chloride (Ag/AgCl) electrode was employed as a non-polarizable gate electrode. All steady-state current-voltage measurements were performed under ambient conditions using an Agilent model E5273A.
A Bruker Dektak XT profilometer with a stylus force of 2 mg was used to measure the film thickness.
Microscope images for the channel geometries were recorded utilizing a Nikon Eclipse LV100 ND microscope in bright-field mode.
Fabrication of ultrathin and biodegradable devices
To create ultrathin flexible devices, a 1.4 µm thick PET (Mylar® 1.4 CW02) foil was adhered on the glass slide. The glass substrates measuring 25.4 × 27 mm were thoroughly cleaned in an ultrasonic bath for 15 min using a series of solutions: a 2% volume solution of Hellmanex detergent in deionized water, followed by deionized water, acetone, and isopropanol. Next, a polydimethylsiloxane (PDMS Sylgard 184 Silicone Elastomer), solution was prepared by mixing the PDMS base and hardener in a 10:1 weight ratio, diluted with n-hexane at a 1:1 weight ratio. The cleaned glass substrates were coated with a thin layer of PDMS at 4000 rpm for 30 s and then annealed at 125 °C for 15 min. This process promotes adhesion between the PET foil and the glass substrate through van der Waals forces. After securely adhering the PET foil to the glass/PDMS surface, the samples were subjected to an additional annealing step at 115 °C for about 15 min to enhance the adhesion of the thin foil. Finally, the glass/PET substrates were cut into three pieces measuring 25.4 mm × 9 mm using a diamond glass cutter.
To prepare the biodegradable OECTs, a 20 µm thick biaxially oriented polylactic acid (BoPLA Folien NTSS NT, Pütz GmbH + Co. Folien KG) film was applied to 25.4 mm by 9 mm glass substrates using double-sided Kapton tape (3MTM). Additionally, the biodegradable molded gelatine wells were used to create fully degradable OECTs.
The OECT devices on PET substrates were completed by applying the same processing steps mentioned above for glass samples. For the OECTs on PLA, the dispersions were drop-cast onto the PLA/glass substrate and then dried and annealed at 100 °C for 20 min, followed by 110 °C for 20. Afterwards, the PET and PLA foils were peeled off and transferred onto a flexible apparatus to measure the device characteristics in initial/compressible and bendable mode. Double-sided acrylic elastomer tapes (3 M VHB) were used as a reliable adhesive supporting platform during device measurements with mechanical strain.
Compression tests were conducted using OECT devices prepared on ultrathin PET foil. To evaluate device performance under initial and compression states, a sticky thick acrylic elastomer (3 M VHB) was used. The tape was fixed in an apparatus where it could be stretched to increase its length and then relaxed. The device was placed onto the tape while it was in a stretched state. This setup allowed the device to be compressed when the tape was relaxed by a certain percentage of its length. Multiple tests were conducted, where devices were measured in their initial state, at 5% compression, and at 10% compression relative to the tape length. The results present the statistical distribution of measurements for at least four devices per condition. Characterization was performed under ambient conditions using an Agilent E5273A.
3D map of compressed PET morphology. The wrinkled morphology of the compressed PET foil was analyzed using a Bruker DektakXT stylus profilometer over a scanned area of 2 × 2 mm with 500 lines of surface mapping.
Tensile tests: Dumbbell-shaped samples of ultrathin PET and PLA were fabricated for uniaxial tensile testing. The tests were conducted at room temperature using a uniaxial tensiometer (Zwick Roell Z005, 100 N load cell) with a 2 mm/min strain rate.
Swelling tests were performed by depositing multiple layers of the biocomposite dispersion with additives as used for the OECT devices via drop-casting. The thick layer of material was then dried for 2 h at 60 °C and then annealed at 120 °C for 30 min. The obtained biocomposite (~20 µm thick) pieces were then put into glass petri dishes and photographed. Afterwards, the pieces were fully immersed in water. Pictures were recorded after 10 min, 1 h, 1 day and 2 days to observe swelling behavior by evaluating geometrical changes. The swelling properties of pure Sacran were investigated by stepwise adding MΩ water (with addition of total water volume of 4 mL, 7 mL, 10 mL, 13 mL, 15 mL, and 20 mL) to the 96 mg Sacran in a crystallization dish. After each additional step, a picture was taken from the swollen Sacran. Finally, after the addition of a total of 20 mL MΩ water, the dish was removed to show the hydrogel structure as well as the absence of liquefaction.
Biodegradation tests of PLA-based OECTs were carried out via enzymatic hydrolysis to significantly accelerate the degradation rate. OECT device sample was placed in a vial filled with 5 mL of phosphate buffer saline (0.05 M, pH 8.0) containing 2 mg of proteinase K and 0.1 mg of sodium azide as an anti-mildew and antibacterial agent. The vials are kept in the reservoir well on a warming plate at 37 °C. The buffer/enzyme system was changed every 48 h to restore the original enzymatic activity67. The weight of the sample was measured periodically on dry basis during the entire process. The sample was withdrawn from the vial at certain time intervals, washed with distilled water, dried, weighed, and then re-immersed into the degradation media. The percentage weight loss (WL%) is calculated by the equation:
$${W}_{L} \% =\frac{\left({W}_{i}-{W}_{f}\right)}{{W}_{i}}\cdot 100$$
(2)
where Wi and Wf are the respective initial and the final mass of the specimen.
In vitro biocompatibility analysis
Thin-film preparation
For the biocompatibility of the materials, 94.5% (v/v) dispersions of PEDOT:Sacran (5:1), PEDOT:Sacran (7.5:1), and PEDOT:PSS were separately mixed with 0.5% (v/v) DBSA and 5% (v/v) glycerol, whereby subsequently 1% (v/v) GOPS was added for adherence on the glass substrates. The glass slides (1 × 1 cm) were cleaned in an ultrasonic bath with a 2% (v/v) aqueous solution of Hellmanex III, deionized water, acetone and isopropanol at 50 °C for 20 min each. The substrates were dried with N2 gas and treated with oxygen plasma at 100 W for 5 min in a Plasma Etch PE-24 plasma cleaner. The respective material dispersions were deposited via spin-coating at 900 rpm for 60 s. The samples were annealed at 140 °C for 1 h for cross-linking GOPS.
Cell culture and cell viability (ISO 10993-5 guidelines)
Cell line L929 (adipose mouse fibroblasts) was purchased from American Type Culture Collection (ATCC, Manassas, VA, USA) and was cultivated in DMEM (Life Technologies, Carlsbad, CA, USA). The culture medium contained (final concentrations in medium): 10% fetal calf serum (FCS), L-Glutamine (2 mM), PenStrep (5 U penicillin, 50 µg streptomycin/mL). Cells were incubated at 37 °C and under a 5% CO2-enriched atmosphere. The LIVE/DEAD assay was performed using a BD FACSVerse flow cytometer (BD Biosciences, San Jose, CA, USA). Heat-killed cells that were incubated at 60 °C for 30 min before measurements were used as a positive control.
After incubation (24 h), we collected the supernatant (500 µL, washed the cells with phosphate-buffered saline (PBS; 0.1 M, pH 7.4; 500 µL), detached the cells with 0.25% trypsin-EDTA solution, and, after 5 min, we resuspended the cells in 750 µL of a culture medium (final volume 2000 µL). There was no volume discarded during the preparation (even PBS for washing was collected), so we obtained information about all cells in our samples. Then, the cells were incubated with PI (10 µg/mL) and calcein-AM (50 µM diluted in DMSO) for 30 min in the dark. Finally, the fluorescence signal was measured on a flow cytometer using the first two scatters (Forward scatter channel vs. Side scatter channel) and then two fluorescence channels (red FL 2: ex. 488/em. 700 nm and green FL 3: ex. 488/em. 527 nm. A red signal due to propidium iodide revealed dead cells that had lost membrane integrity, whereas a green signal indicated live cells with active intracellular esterases that could catalyze the non-fluorescent calcein-AM to highly fluorescent green calcein. Three independent experiments were performed and mean and standard deviation ± (SD) were calculated.
link





More Stories
Researchers Reveal Molecular Secrets of Flexible Electronics
Growth Outweighs Uncertainty for Flexible and Printed Electronics: OE-A Survey
SEMI FlexTech Announces 2026 FLEXI Award Winners