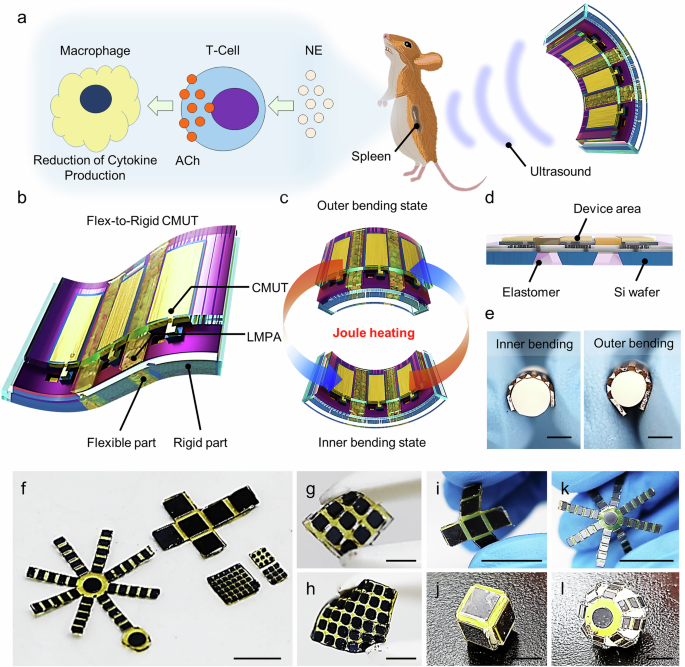
Fabrication process of flex-to-rigid structures
To form flex-to-rigid (FTR) structures, the double-sided polishing (DSP) silicon substrate (Unisill Co., Korea) was used for the rigid parts, and LMPA and photo-sensitive polyimide (PI; GPI-102K, PNS technology, Korea) were used for the flexible regions bridging the rigid parts. For the fabrication of silicon islands, thermal oxidation was performed at 1050 °C for 6 min on a DSP silicon wafer using a furnace (E1200, Centrotherm, Blaubeuren, Germany). The backside oxide layer was first patterned using a photoresist and a buffered oxide etch (BOE; Sigma-Aldrich, Saint Louis, USA). Before etching the backside silicon layer, a 50-μm-thick polydimethylsiloxane (PDMS) was coated and cured using a vacuum oven at 85 °C for 4 h on the front side of the Si wafer to protect the front silicon. The backside silicon was anisotropically etched using 5% tetramethylammonium hydroxide (TMAH; Sigma-Aldrich, Saint Louis, USA) with hydroxylamine (NH2OH; Sigma-Aldrich, Saint Louis, USA). The volume ratio of the solution was TMAH:NH2OH = 20:1. After etching the bottom silicon, the PDMS was removed using propylene glycol monomethyl ether acetate (PGMEA; Sigma-Aldrich, Saint Louis, USA) with tetrabutylammonium fluoride solution (TBAF; Sigma-Aldrich, Saint Louis, USA). Next, the wafer was dipped into the BOE solution while covering the backside using a wafer holder to protect the backside SiO2 mask layer (Supplementary Fig. 21). The volume ratio of the solution was PGMEA:TBAF = 99:1. In order to coat polyimide on the frontside wafer, the dummy wafer was bonded to the bottom side using a crystal adhesive bond (821-3 CrystalbondTM 509 Clear, TED PELLA. INC., Redding, USA). A 3-μm-thick photo-sensitive polyimide was coated using a spin coater at 1000 rpm for 20 s and baked at 110 °C for 2 min. The polyimide was exposed using a contact aligner (UV exposure, 200 mJ, 365 nm) and developed in AZ300MIF for 1 min. The patterned polyimide was cured at 250 °C for 2 h. Deep Reactive Ion Etching (DRIE) was performed to remove the remaining thin silicon present in the trench between the rigid islands, and SiO2 acted as the etch stop layer. PDMS was poured on the trench area, and saw dicing was conducted to define each FTR structure.
Fabrication process of flexible FTR CMUTs
For the fabrication of flexible CMUTs in the FTR structures, we started with a highly-doped (boron) double-sided Si wafer ( < 0.005 Ω∙cm) and silicon-on-insulator (SOI) wafer (500-μm-thick device layer (Si), 2-μm-thick box layer (SiO2), and 725-μm-thick handling layer (Si)) (Supplementary Fig. 4). First, 100-nm-thick insulation layers were grown on the Si and SOI wafers through the furnace (wet oxidation at 1050 °C). Next, the vacuum cavities were defined by patterning the thermal oxide layer on the SOI wafer using a 1-μm-thick PR and BOE solution (etch time of 1 min 22 s). To enclose the vacuum cavity with top silicon membranes, the Si wafer and SOI wafer were bonded at the oxide interfaces in a high vacuum using a wafer bonder (SB8 GEN2, SÜSS MicroTec, Garching, Germany). Prior to the wafer bonding, a cleaning step of SPM (H2SO4:H2O2 = 3:1 vol%) at 70 °C was conducted for 10 min followed by SC-1 (H2O:H2O2:NH4OH = 5:1:1 vol%). In order to form a strong bonding at the oxide interface, annealing was conducted in a furnace at 1050 °C in a nitrogen ambient for at least 2 h. The uniform bonding at the interface between oxide layers was confirmed through scanning acoustic microscopy (SAM; Gen6TM C-SAM, Santa Clara, USA). The patterned CMUTs were observed as grey rectangular shapes, and a few defects were observed as black circular and oval shapes (Supplementary Fig. 5). After the wafer bonding, the handling layer was removed by grinding, chemical mechanical process (CMP), and Si wet-etching using TMAH (5%) with NH2OH solution. Next, the silicon islands were created using the fabrication process described in the previous session. To prevent fracturing of the wafer, a 4-inch dummy Si wafer was attached to the bottom using a crystal adhesive bond.
The top silicon was then patterned by the reactive ion etching after patterning with a photoresist to define CMUT membranes. After patterning the membrane, the PR was stripped in the PR stripper (APPS-1) at 50 °C for 10 min. The vias to the ground were formed by etching the top oxide layer using reactive ion etching (RIE). After the elimination of the native oxide layer on the membrane, 10 nm chrome (Cr) and 100 nm gold (Au) layers were deposited using the thermal evaporator to form signal and ground pads. In the same manner as the method of forming the FTR structure, photosensitive polyimide having a thickness of 3 μm was coated and patterned between CMUT elements. After attaching a dummy wafer to the front of the wafer through a crystal bonder to remove the Si layer remaining in the rear, the DRIE process was performed, and SiO2, which had been grown for the insulation layer, was used as the etch stop layer. After the DRIE process, the saw dicing separated the part corresponding to each area of the device with the dummy wafer attached. LMPA was coated on the etched area using a micro syringe needle, at which time the LMPA was placed on a hot plate of 60 degrees or more to be transferred in liquid form. After the LMPA changes to a solid state, soak it in acetone for 30 minutes to detach the dummy wafer by removing the crystal bonder, and clean the IPA and DI for 3 minutes to remove the residue remaining on the surface. To protect against the leakage of LMPA during the bending operation, PDMS was molded to the backside of the CMUT while the front side of the CMUT was protected by water-soluble tape. The PDMS was cured in a vacuum oven at 85 °C for 2 hours, and then to remove water-soluble tape, the device was submerged in the DI for 1 hour. The fabricated FTR CMUTs were connected electrically to the flexible printed circuit board (FPCB) through the wire bonding process (Supplementary Fig. 22). Finally, the CMUTs integrated with the FPCB were dip-coated in a PDMS solution using a dip coater at a coating speed of 0.05 mm/s for encapsulation.
CMUT design
We used the finite element method (FEM)-based COMOSOL simulation to design a single CMUT cell. For analytical calculations, we used the equations shown below to determine resonant frequency and pull-in voltage:
$${f}_{0}=0.469\frac{{t}_{m}}{{R}_{m}^{2}}\sqrt{\frac{\frac{E}{{\rho }_{1}(1-{\sigma }^{2})}}{1+0.67\frac{{R}_{m}{\rho }_{2}}{{\rho }_{1}{t}_{m}}}}\propto \sqrt{\frac{{t}_{m}^{3}}{{R}_{m}^{5}}}$$
$${V}_{{pull}-{in}}=7.542\sqrt{\frac{{g}_{0}^{3}D}{{R}_{m}^{4}{\varepsilon }_{0}}}\propto \frac{{g}_{0}^{\frac{3}{2}}}{{R}_{m}^{2}}$$
where \({R}_{m}\) is the radius of the cavity, \({t}_{m}\) is the thickness of the membrane, \(E\) is Young’s modulus of the membrane, \({\rho }_{1}\) is the density of the membrane, \({\rho }_{2}\) is the density of a surrounding liquid, \(\sigma\) is Poisson’s ratio of the membrane, \({g}_{0}\) is the gap height of the cavity, \({\epsilon }_{0}\) is the vacuum permittivity, and \(D\) is the flexural rigidity of the membrane. The materials used for the membrane, insulation layer, and electrode were Si, SiO2, and Cr/Au, respectively. The designed single CMUT cell consisted of a circular cell with a radius of 11 μm, a 450-nm-thick membrane, and a 100-nm-thick vacuum gap height. The expected pull-in voltage was 60 V, the center frequency was 8.67 MHz in air, and the maximum pressure in water was 175 kPa (Supplementary Fig. 23, Table 1).
Mechanical characterization
The bending tests of the FTR CMUTs were conducted using a universal testing machine (USM-500N, A&D CO., LTD, Japan). The short edges of the FTR CMUT were attached to glass slides which were clamped to the holders in the universal testing machine. The bending speed was 30 mm/min. The FTR CMUT was bent up to the radius of curvature of 2.5 mm at a constant rate. For the fatigue bending test, 100 bending cycles were performed at a constant bending speed in both inner and outer bending with a radius of curvature of 2.5 mm.
Electrical impedance characterization
The electrical properties of CMUTs were analyzed using an impedance analyzer (E4990A, Keysight Technologies, USA) in air. A bias tee consisting of a 1 MΩ resistor and a 22 nF capacitor connected in parallel was used to superimpose the DC voltage on AC voltage during the measurement of impedance in CMUT. By increasing the DC bias voltage in step of 5 V using the high voltage supply (PS310, Stanford Research Systems Inc., Sunnyvale, CA, USA), the operational voltage (80% of collapsed voltage) was confirmed. The components in the equivalent circuit (6-component model) of CMUT were extracted from the impedance analyzer.
Acoustic characterization
To observe the movement of the membrane on elements, the laser Doppler vibrometer (LDV; FV-534, OFV-2570, Polytec GmbH, Germany) was utilized (Supplementary Movie 1). To measure acoustic properties, the FTR CMUTs connected to the FPCB were passivated with PDMS to prevent an electrical shortage of the device in water. The FTR CMUTs were actuated using a function generator (33220A, Agilent Technologies, CA, USA) and an RF amplifier (BT00100-AlphaS-CW, Tomco Technologies, Australia). The generated AC signal was superimposed on a DC signal through a bias tee. The ultrasound generated from the FTR CMUTs was measured using a needle-type hydrophone (NH0500, Precision Acoustics, UK), which was fixed on a programmable motorized stage (Sciencetown Inc., Incheon, Korea). To accurately determine the focal point, three-dimensional scan beam profiling was performed at intervals of 250 micrometers, focusing on a section extending 24 mm from the CMUT surface. The acoustic wave signals were recorded using a high-speed oscilloscope (DSOX2022A, Agilent Technologies Inc., CA, USA).
Bending modulation via joule heating
To confirm the bending modulation based on the phase transition of LMPA via joule heating, a lower voltage than 5 V was applied to the ground pads at both ends of the FTR CMUTs to induce Joule heating. The temperature generated by Joule heating was directly measured by inserting the thermocouple (AT-3K, Hanyoungnux, Korea) into the passivated liquid metal. The phase transition time was confirmed through an optical microscope based on the degree of hardening of the liquid metal.
Collagen emulsion formation
For the formation of a collagen emulsion, we used an immunization-grade chick type II collagen (20012, Chondrex Inc., USA), a Complete Freund’s adjuvant (CFA, 7001, Chondrex Inc., USA), a 3-way stopcock, a 5 ml syringe, a homogenizer (Omni TH Tissue Homogenizer, OMNI International, USA) with a small blade (diameter of 5 mm), and an ice water bath to maintain the emulsion in a cold state during mixing. First, the CFA was added to the syringe, which was sealed with a 3-way stopcock, and then the collagen solution was added gradually at an equal volume of CFA. The emulsion was mixed at 30,000 rpm for 2 minutes before being cooled in an iced water bath for 5 minutes. The mixed collagen emulsion was then dropped into the water to verify that the emulsion was formed correctly (Supplementary Fig. 24).
Collagen-induced arthritis animal model
All animals used in this study were in accordance with protocols outlined and approved by the Institutional Animal Care and Use Committee at the Korea Advanced Institute of Science and Technology (KA2022-022). For the development of CIA, DBA/1 J type (male, 6~7 weeks) mice (n = 31), which are genetically susceptible to autoimmune diseases, were used. The mice were habituated in the animal cage in our lab for a week before the injection of collagen emulsion. Mice were anesthetized with isoflurane (4% induction, 2% maintenance at an O2 flow rate of 1 L/min) using an isoflurane vaporizer system (RWD Life Science Co., Ltd., China), and the 100 μL collagen emulsion was injected subcutaneously into the tail with the tip of the needle inserted between 0.5 and 2 cm from the base of the tail using a 22-gauge Hamilton syringe (1 mL, Gastight Syringe Model 1001 RN, Hamilton®, USA) (Supplementary Movie 2). After 3 weeks from the injection of collagen emulsion, rheumatoid arthritis (RA) was observed through the swelling of ankles. After 6 weeks from the injection, RA was expressed in all of the injected mice (Supplementary Fig. 25a).
Measurement of ankle thickness
To quantify the thickness of swollen ankles due to the expression of rheumatoid arthritis, the thickness of both forelimbs and hindlimbs of 31 mice was measured using a digital thickness gauge (BD547-301, Blue Tec, Korea) every week over 8 weeks. Each ankle was measured three times and the values were averaged. To obtain exact ankle thickness on the same location, the mice were anesthetized with isoflurane (4% induction, 2% maintenance at an O2 flow rate of 1 L/min) on a temperature-controlled warming pad (RT-0515, Kent Scientific Corporation, USA) which was maintained at 37 °C during the measurements (Supplementary Fig. 25b).
Clinical score of rheumatoid arthritis
The thickness of mice ankles was measured for 8 weeks, and each paw was assigned a clinical score for the degree of inflammation from 0 to 4 refers to the scoring system introduced by Chondrex, Inc. (www.chondrex.com/documents/Scoring-System.pdf). For each paw, the clinical score was designated 0 for no erythema and swelling, 1 for mild erythema and swelling that was restricted to the tarsals or ankle joint, 2 for moderate erythema and swelling that extended from the ankle to the metatarsal joints, 3 for severe erythema and swelling that covered the ankle, foot, and digits, or 4 for ankylosis. The maximum scale of clinical scores, adding all paws, was 16 (Supplementary Fig. 25c).
In vivo ultrasound stimulation
For analysis of the effect of the ultrasound stimulation, mice were classified into four groups (stimulation, sham, model, and control cohorts). The sham cohort was subject to the same setup as the stimulation cohort but without ultrasound stimulation. The FTR CMUT integrated with the flexible printed circuit board (FPCB) was positioned on the dorsal skin with ultrasonic gel, slightly lateral to the spinal cord (Supplementary Fig. 26). The ultrasound generated by the FTR CMUTs stimulated the spleen located beneath the dorsal skin (Supplementary Fig. 27). All channels of the CMUT were activated simultaneously by wire-bonding each channel to common signal and ground pads on the FPCB. All channels were biased at a DC voltage of 80 V and were driven by a sine wave at a frequency of 1 MHz with an AC voltage of 17.67 V. The ultrasound with a peak pressure of 350 kPa was delivered for 20 minutes per day (pulse repetition frequency: 0.167 Hz, duty cycle: 16.7%). This treatment was administered 3 times a week for 2 weeks, 6 weeks after the collagen injection. For comparison of the thickness and degree of rheumatoid arthritis, the thickness of all ankles was measured every week.
Gait analysis
A custom-made gait analysis system was used in this experiment. An enclosed walkway consisted of a horizontal glass bottom plate with a blacked-out tunnel on the sides and top, allowing mice to walk through. A green LED light strip was attached to a long edge of the glass bottom plate using Loctite super glue. Black-colored tape was used to cover the opposite long edge of the glass plate to ensure the internal reflection of the light did not escape. A web camera (TC70, TP-Link, China) placed underneath the glass walkway was used to monitor the footprints. Each mouse was placed at the opening of the tunnel to complete three uninterrupted runs along the glass walkway. The experiment was conducted in a dark chamber for conducive mice locomotion. The video was recorded in night view mode with a frame rate of 30 frames/second for behavioral analysis.
DeepLabCut analysis setup
To analyze the gait of the mice in the recorded videos, we utilized DeepLabCut (DLC), a versatile and efficient machine-learning (ML)-based toolbox for animal pose estimation. We employed a GPU-based version of DLC on a Linux system (Ubuntu 16.04), equipped with a 32GB AMD Ryzen 7 1700 8-core CPU and NVIDIA Titan XP GPU. To train our model, we manually labeled each paw and toe for a total of 22 points per mouse as well as 4 reference points from 26 representative videos across 4 batches of experimental data. The ResNet-50 convolutional neural network (CNN) was used to train the system for 800,000 iterations, and the trained DLC network autonomously labeled all the extracted frames from a total of 148 experimental video recordings. The resulting trajectory data was analyzed using a custom-coded MATLAB program to calculate gait characteristics such as the frequency and width of footprints in contact with the glass walkway, and the stride length for each paw and toe (Mathworks®, Natick, MA, USA). The unit for distance was converted into centimeters using the ratio between the actual width of the glass plate and the distance in pixels between the reference points in the video.
Statistical analysis
Statistical analysis was conducted on normally distributed data sets confirmed using Shapiro–Wilk’s normality test. Parametric analysis was performed using a two-sided unpaired Student’s t-test (unequal variances) for behavioral data, and one-way analysis of variance (ANOVA) with Fisher LSD’s post-hoc test for ultrasound treatment data. For one-way ANOVA, the experimental group was used as the factor, and the stimulation, sham, and model groups were used as factors. All data were analyzed using Microsoft Excel (Microsoft) and OriginPro 2019 (OriginLab Co., Northampton, MA, USA).
link





More Stories
Researchers Reveal Molecular Secrets of Flexible Electronics
Growth Outweighs Uncertainty for Flexible and Printed Electronics: OE-A Survey
SEMI FlexTech Announces 2026 FLEXI Award Winners