High-performance OECTs based on electropolymerized small-anion doped PEDOT
The main challenge in fabricating electropolymerized OECTs is depositing the channel material onto insulating substrates. This can be addressed by short-circuiting metal source-drain electrodes, spaced by a moderately small gap (typically less than 50 µm), and using them as a single working electrode during electropolymerization43. This setup enables the formation of a continuous channel by bridging the insulating gap between the electrodes with the electropolymerized material. Other approaches include using alternating current to promote fiber formation along the direction of the electric field, eventually creating dendritic networks44,45, and precoating the channel area with solution-processed PEDOT46,47.
We electropolymerized PEDOT:ClO4 films onto short-circuited source-drain electrodes patterns to fabricate OECTs (Fig. 1a) using varying concentrations (1, 5, 100 mM) of the dopant LiClO4. All devices exhibited p-type behavior and operated in depletion mode, as expected for PEDOT-based OECTs18. Notably, electrolyte concentrations of 5 mM and 100 mM both resulted in complete channel coverage, yielding OECTs with similar electrical characteristics (Supplementary Fig. 1). In contrast, a 1 mM concentration was insufficient to bridge source and drain electrodes, as no significant current was detected during electrical measurements. We consistently used high concentration of 100 mM in this study, as it is commonly employed in electrochemical systems for its high ionic conductivity and reduced ionic migration48.

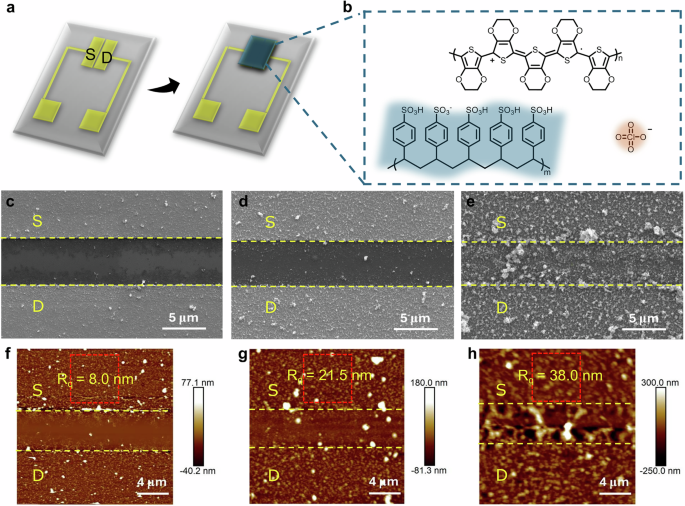
a Schematic illustration of the OECT structure with patterned Au source (S)-drain (D) electrodes and electrodeposited PEDOT channel film. b Chemical structure of PEDOT+, PSS− and ClO4−. SEM (c–e) and AFM (f–h) images of PEDOT:ClO4 films electropolymerized at charge densities of: c, f 20 mC/cm2, d, g 30 mC/cm2 and e, h 60 mC/cm2. Yellow dashed lines: OECT channel. Red dashed box: region from which the surface roughness Rq is extracted.
The morphology of PEDOT:ClO4 films electrodeposited at charge densities of 20 (Fig. 1c, f), 30 (Fig. 1d, g), and 60 (Fig. 1e, h) mC/cm2 was investigated using scanning electron microscopy (SEM) and atomic force microscopy (AFM). As expected, electropolymerization initiated at the Au electrodes and progressively extended across the channel area, with growth occurring both laterally and vertically relative to the electrode surface. This behavior aligns with established nucleation-growth mechanisms, resulting in a progressive increase in surface roughness (root mean square roughness, Rq) at longer deposition times49. At an early stage (20 mC/cm², 40 s), the film primarily formed on the source and drain electrodes, with limited extension into the adjacent channel regions. The resulting morphology displayed an Rq of 8 nm (Fig. 1c, f). Increasing the deposition charge (30 mC/cm², 60 s) led to full channel coverage, yielding a granular network on the Au electrodes (Rq = 21 nm, Fig. 1d, g; Supplementary Fig. 2a) and densely packed aggregates across the channel (Supplementary Fig. 2b). Further increasing the deposition charge (60 mC/cm², 120 s) led to continued film growth atop the existing PEDOT layer, producing a rougher granular morphology across both the electrodes and channel (Rq = 38 nm, Fig. 1e, h; Supplementary Fig. 2c). Although the morphological image does not show a complete connection between the source and drain electrodes at low deposition charge (20 mC/cm2, Fig. 1c, f), localized conductive pathways may lead to devices showing a low source-drain current and a limited performance in terms of transconductance (0.1 mS) and ON/OFF ratio (2 × 101) (Supplementary Fig. 3a, d). When the channel was fully covered, at 30 mC/cm2 (Supplementary Fig. 3b, e) and 60 mC/cm2 (Supplementary Fig. 3c, f), the devices displayed similar performance with high transconductance (above 10 mS), and an ON/OFF ratio around 1 × 103, despite minor changes in morphology (Supplementary Fig. 2). Electrodeposited OECTs obtained with other small dopants (Supplementary Fig. 4), such as LiBF4 and TEABF4, frequently used for PEDOT electrodeposition, showed similar transconductance (~10 mS) and ON/OFF ratios (~4 × 102), indicating that various combinations of small anion dopants and accompanying cations do not significantly impact the electropolymerization process, consistent with previous findings50. Overall, our results show that electrodeposition produces highly reproducible devices with performance comparable to state-of-the-art electropolymerized OECTs (Supplementary Table 1).
Devices with similar steady-state characteristics (Fig. 2) were obtained using the larger polymeric dopant polystyrene sulfonate (PSS−), both through electrodeposition (gm~10 mS, ON/OFF ratio ~6 × 102) and inkjet printing from a commercial suspension (gm~9 mS, ON/OFF ratio ~7.4 × 102) (Table 1). Notably, the drain current (ID) saturated at ~VG = 0 V for PEDOT:ClO4 OECTs (Fig. 2a, b, and Supplementary Fig. 5a, d), but at a slightly negative VG for the electropolymerized (Fig. 2d, e, and Supplementary Fig. 5b, e) and printed (Fig. 2g, f, and Supplementary Fig. 5c, f) PEDOT:PSS OECTs. This indicates that, at VG = 0 V, PEDOT:ClO4 films are fully doped, while PEDOT:PSS films are partially doped. This is further supported by UV-Vis spectroscopic measurements, which are discussed in detail in the following section. Therefore, PEDOT:ClO4 devices can operate with lower energy consumption. These findings demonstrate that the ClO4− dopant promotes a fully doping state in PEDOT films, likely due to the more efficient insertion and redistribution of the smaller dopant ions during electrodeposition. Remarkably, all devices retained similar performance after 4 months of storage in ambient conditions (Fig. 2b, e, h).
a–c electropolymerized PEDOT:ClO4 with deposition charge density of 60 mC/cm2; d–f electropolymerized PEDOT:PSS with deposition charge density of 300 mC/cm2; g–i ink-jet printed PEDOT:PSS. A Ag/AgCl pellet gate and 100 mM NaCl electrolyte were used in all measurements. Transfer curves were measured at a VD = −0.4 V. For all fabrication conditions, three devices were measured to confirm their reproducibility. All samples were measured one day after fabrication. The shaded area around the transfer curves represents the standard deviation extracted from three samples.W/L = 4000 µm/5 µm.
Despite their similar steady-state performance, the devices exhibited different stability under cyclic pulsed VG application. To assess cycling stability, we monitored current responses during gate voltage pulses (VG = 0.8 V, pulse width = 5 s) over time. For PEDOT:ClO4 OECTs, the current (ID) remained nearly constant, with only a 2% decrease after 1000 cycles (Fig. 2c). Electrodeposited PEDOT:PSS-OECTs showed a roughly 20% decline in ID (Fig. 2f). A larger ID drop of about 50% was observed in printed devices (Fig. 2i). The reduced cycling stability of PEDOT:PSS is primarily attributed to the greater swelling of its hydrophilic PSS chains51,52,53. In inkjet-printed devices, this effect is more pronounced due to the excess PSS required to stabilize commercial PEDOT:PSS dispersions. The higher PSS content increases film hydrophilicity, leading to enhanced swelling in aqueous environments and reduced operational stability. This observation is consistent with our previous findings: solution-processed PEDOT:PSS films showed a significant weight increase after 10 min of water immersion, whereas PEDOT:ClO4 films exhibited minimal change54. Beyond swelling, differences in channel/electrode interface quality, arising from distinct fabrication methods, may also contribute to the observed performance variations. Overall, electropolymerized OECTs demonstrated superior stability compared to printed devices, particularly when the small dopant anion ClO4− was used. While inkjet printing is widely used for fabricating flexible electronic devices, the advantages of electropolymerization underscore its promising potential for the development of flexible OECTs.
Electropolymerized PEDOT:ClO4 on parylene substrate for flexible and lightweight OECTs
Flexible and lightweight OECTs were fabricated by electropolymerizing PEDOT:ClO4 films onto source-drain electrodes patterned on a thin (10 μm) parylene layer deposited on a glass substrate. At the end of the fabrication process, the parylene layer was peeled off from the glass to yield ultrathin flexible devices (Fig. 3a). Compared to conventional flexible substrates such as polyimide or polyethylene terephthalate (PET), ultrathin parylene films significantly reduce overall device weight and allow conformability to small-radius curved surfaces, facilitating mechanical flexibility testing. The ability of a material to conform to curved surfaces is governed by its bending stiffness, which is proportional to the Young’s modulus (E) and the cube of its thickness (t³). For a given material, where the Young’s modulus is constant, reducing the thickness significantly lowers the bending stiffness, allowing it to bend more easily and conform to surfaces with smaller radii of curvature. In the flat state, the flexible devices exhibited a transconductance of 7.6 mS and an ON/OFF ratio of 2.5 × 102 (Fig. 3b), slightly lower than those of the rigid counterparts, likely due to variations in PEDOT film growth influenced by differences in gold electrode surfaces across substrates (Supplementary Fig. 6). Importantly, the transfer characteristics remained nearly unchanged under mechanical deformation applied either parallel (Fig. 3c) or perpendicular (Fig. 3d) to the channel length, demonstrating robust flexibility in both directions. After recovery from bending, the devices retained a transconductance of 7.4 mS and an ON/OFF ratio of 2.3 × 102, confirming stable performance after mechanical strains. Further evaluation of mechanical durability and long-term reliability in future will be important for advancing their practical application in flexible electronics.
a Schematic illustration of the flexible OECT. Transfer characteristics of the flexible OECT measured b before mechanical bending and after recovery from mechanical bending; c at the flat state and under bending parallel to the channel length; d at the flat state and under bending perpendicular to the channel length. (Insets: photographs of the device bending along different directions on a glass rod with the diameter of 5 mm).
Electrochromic behavior of PEDOT:ClO4 films during doping/dedoping monitored by UV–Vis spectroelectrochemistry
UV–Vis spectroelectrochemistry has been widely used to study changes in doping state of conducting polymers during electrochemical doping and dedoping processes55. For our study, we monitored the UV–Vis absorption spectra of electrodeposited PEDOT:ClO4 films in real time while applying cyclic linear voltage sweeps between −1.0 and 0.4 V, effectively capturing the dedoping (Fig. 4a) and redoping (Fig. 4b) processes. Consistent with prior studies on PEDOT films56, we observed a characteristic absorbance band in the 400–700 nm range, which corresponds to the π–π* transitions of PEDOT neutral chains. Additionally, a second band appeared in the near-infrared range (700–1100 nm), associated with polaron absorption. The bipolaron states, which are typically located in the far infrared range, were not captured in our spectra. To analyze these changes more closely, we monitored selected wavelengths over time (Fig. 4c lower panel): the 621 nm curve (blue line), representing neutral chain signatures, increased significantly as the potential shifted from 0.4 to −1.0 V (Fig. 4c top panel). Simultaneously, the 801 nm (pale red line) and the 1000 nm (red line) curves, corresponding to the polaron band, decreased, consistent with the dedoping of the polymer film. During the redoping process, within the polaron absorption region (800 nm and 1000 nm), a unique behavior was observed near the highly doped state (around 0.4 V). The 800 nm peak displayed a dip, while the 1000 nm peak reached maximum intensity. In contrast, the 621 nm signal continued to decrease. These features suggest that, while the number of neutral chains decreases during redoping, the polarons do not increase to their maximum concentration but instead transition into other species, possibly bipolarons. This observation aligns with the idea that, under highly doped conditions, some polarons convert to bipolarons, indicating complex dynamics in the electronic states of PEDOT under different electrochemical conditions.
Real-time absorption spectra during a dedoping (+0.4 to −1.0 V) and b redoping (−1.0 to +0.4 V) presented with 0.1 V increments. c top layer: voltage sweep, lower layer: temporal behavior of the three wavelengths chosen as representative of neutral chains (621 nm), polarons (801 nm), and polarons + bipolarons (1000 nm).
Similar results were observed for films electrodeposited from a 5 mM solution (Supplementary Fig. 7), indicating that dopant concentration has minimal impact on the electrical modulation of electropolymerized PEDOT films. This finding aligns with the comparable performance observed in OECTs, reinforcing that variations in dopant concentration do not significantly influence the electronic properties and functionality of these films.
UV–Vis spectroscopic comparison of PEDOT:ClO4 and PEDOT:PSS at 0 V bias highlights distinct differences in their doping states (Supplementary Fig. 8). PEDOT:PSS displays a pronounced absorption band between 600 and 800 nm (black shaded area), while PEDOT:ClO4 exhibits stronger absorption beyond 900 nm (red shaded area). The latter is indicative of increased bipolaron formation, as previously discussed, and reflects a higher doping level in the electropolymerized PEDOT:ClO4 film.
Different ionic injection/ejection behaviors for PEDOT:ClO4 and PEDOT:PSS measured by EQCM-D
To further investigate the doping and dedoping mechanisms in the devices, we employed Electrochemical Quartz Crystal Microbalance with Dissipation (EQCM-D) to monitor in situ mass changes of the channel materials under applied bias in aqueous NaCl. The changes in resonance frequency (Δf/n) and dissipation (ΔD) across multiple harmonics were recorded during cyclic linear potential sweeps, allowing us to gain insight into the dynamic mass uptake and release associated with electrochemical modulation. This approach provides valuable information on how mass transport correlates with the electronic state changes within the channel materials during the doping and dedoping processes, for both rigid and flexible devices.
For quantitative mass analyses, the mass variation (Δm) at the sensor surface or within the deposited film can be determined from the recorded oscillation frequency change (Δf) across different overtones orders (n) using the Sauerbrey’s equation (Eq. 1):
$$\Delta {\rm{m}}=-\frac{{{\rm{Z}}}_{{\rm{q}}}}{2{{\rm{f}}}_{0}^{2}}\times \frac{\Delta {\rm{f}}}{{\rm{n}}}=-{\rm{C}}\times \frac{\Delta {\rm{f}}}{{\rm{n}}}$$
(1)
where Δf/n is the change of frequency at nth overtone. C = Zq/2f02 is the calculation constant (or sensitivity factor) of the quartz crystal, where f0 is the fundamental resonance frequency, and Zq is the acoustic impedance of the quartz crystal (~8.8 × 105 g/cm2s). For a 5 MHz crystal sensor, C equals 17.7 ng/cm2Hz57.
According to the Sauerbrey equation, an increase in Δf/n corresponds to a mass loss, and a decrease corresponds to a mass gain. This equation is typically applicable to rigid materials, such as metal coatings, in air or liquid58. For polymer coatings in contact with liquid media, the Sauerbrey model remains valid if the dissipation change is minimal (ΔD ≈ 0); otherwise, viscoelastic models are preferable58. It has been shown that for polymer films thinner than 100 nm, the Sauerbrey model reliably reflects mass changes (Δm)40,41. When dissipation changes are small, the frequency shifts (Δf/n) overlap across all measured harmonics, indicating that the film behaves as a rigid layer firmly attached to the sensor surface59. In our experiments, all electrodeposited films showed negligible shifts of Δf/n and ΔD over a complete cycle, spanning voltage sweeps from −0.8 to +0.8 V and back (Supplementary Figs. 9, 10). This behavior suggests minimal ionic accumulation or loss, indicating stable mass dynamics during the electrochemical modulation processes.
The EQCM-D responses for PEDOT:ClO4 and PEDOT:PSS films deposited over a 30-s period showed negligible changes in ΔD, along with overlapping Δf/n values across different overtones (n = 3, 5, and 7) (Supplementary Fig. 9a, b), indicating that these films could be reliably analyzed for mass transport. The cyclic voltammograms (CV) for PEDOT:ClO4 (Fig. 5c and Supplementary Fig. 11c) exhibited the typical shape of CV for PEDOT60, with increasing current at faster scan rates, suggesting a pseudo-capacitive behavior. A similar CV profile was observed for PEDOT:PSS (Fig. 5e). The peak observed at −0.4 V, which diminishes upon degassing (Supplementary Fig. 12), is related to oxygen electrochemistry under normal operating conditions.
Voltage sweep, current response, frequency response (Δf/n) at the 3rd overtone, and converted mass variation (Δm) under cyclic linear potential sweep for a PEDOT:ClO4 and b PEDOT:PSS. The potential was applied between −0.8 V to 0.8 V at a scan rate of 50 mV/s. c Cyclic voltammetry, d mass variation vs. voltage for PEDOT:ClO4. e Cyclic voltammetry, f mass variation vs. voltage for PEDOT:PSS. Illustration of the ionic distribution without voltage application and during redoping/dedoping g for PEDOT:ClO4 and h for PEDOT:PSS. The bottom yellow part indicates the Au QCM sensor. For (a and b), the data from the 5th and 6th cycles are shown, and for (c–f), the 5th cycle is plotted.
Before a full cycle sweep, the PEDOT films were held at −0.8 V to achieve a fully dedoped state, due to the injection of the Na+ ions from the electrolyte and, where feasible, the ejection of mobile dopant anions. During the subsequent voltage sweep from −0.8 to 0.8 V (redoping), electrolyte cations migrate toward the counter electrode, while anions migrate to the PEDOT working electrode and enter the film. The ionic movement occurs oppositely during the reverse sweep from 0.8 to −0.8 V (dedoping).
The mass changes for PEDOT:ClO4 during CV were determined from the frequency responses at the 3rd overtone across different voltage scan rates: 20 mV/s (Supplementary Fig. 11a), 50 mV/s (Fig. 5a), and 100 mV/s (Supplementary Fig. 11b). Illustrations of the ionic distribution at the initial state (without voltage applied), during redoping (from –0.8V to 0.8 V) and during dedoping (from 0.8V to –0.8 V) are provided for PEDOT:ClO4 (Fig. 5g) and PEDOT:PSS (Fig. 5e). These mass variations correlated closely with the voltage and current profiles at all scan rates, with only minor differences observed (Supplementary Fig. 11d), indicating that ionic movement is minimally impacted by scan rate. For PEDOT:ClO4, redoping (Fig. 5a, pink shaded area, and Fig. 5d, pink line) begins with a decrease in Δm between −0.8 to −0.3 V due to the ejection of Na+ ions (Fig. 5g). Beyond −0.3 V, Δm sharply increases as ClO4− dopant anions (Fig. 5g), previously ejected during dedoping, re-enter the film possibly accompanied by Cl− anions from the electrolyte. During the reverse sweep (dedoping), Δm continuously decreases (Fig. 5a blue shaded area, and Fig. 5d, blue line), indicating that ClO4− and Cl− anions leave the film as Na+ ions are injected (Fig. 5g). PEDOT:ClO4 samples deposited for 60 s and 120 s exhibited larger ΔD values and pronounced Δf/n splitting across different overtones n = 3, 5 and 7 (Supplementary Fig. 10), suggesting that the Sauerbrey equation is no longer applicable. It has been reported that higher-order shear waves are more sensitive to the material near the electrodes, while lower-order waves experience less damping in the film. Frequency shifts were smaller at higher-order overtones and larger at lower-order overtones, indicating that mass changes are more pronounced closer to the electrolyte. This observation differs from the case of acid-treated PEDOT:PSS, which has been reported to exhibit greater mass change near the substrate39.
For PEDOT:PSS, during redoping, a substantial mass decrease was observed (Fig. 5b pink shaded area, Fig. 5f pink line), indicating that the process is primarily driven by the ejection of Na+ cations from the film, along with the injection of Cl− anions (Fig. 5h). During dedoping, Δm remained stable from 0.8 to 0.4 V but increased between 0.4 and -0.8 V (Fig. 5b blue shaded area, Fig. 5f blue line). The stable Δm between 0.8 and 0.4 V suggests a mass balance between the slight ejection of Cl− ions and the injection of Na+ ions. The subsequent mass increase indicates that the process is dominated by Na+ ion injection to compensate negatively charged PSS chain (Fig. 5h), consistent with the widely accepted understanding of the doping/dedoping mechanism in PEDOT:PSS. Unlike previous findings regarding spin-coated PEDOT:PSS films containing additives40, our results demonstrate the participation of Cl− anions in this process.
Therefore, EQCM-D enabled a direct evaluation of the doping/dedoping mechanisms in electropolymerized PEDOT films, revealing that small anion-doped PEDOT (PEDOT:ClO4) differs from polyanion-doped PEDOT (PEDOT:PSS) due to the distinct involvement of dopant counterions. While EQCM-D offers valuable insights into mass transport during electrochemical processes, it presents limitations on resolving contributions from various ionic species, such as Na+ and ClO4−, and from the solvent. As such, a more detailed understanding of mass transport will require the integration of advanced operando characterization techniques, such as XRF for substituted ions.
link