
Metallization and characterization
Magnolia liliiflora leaves were extracted locally and the mesophyll was removed using an alkaline etching process based on previous report4in order to expose the internal quasi-fractal vasculature of the leaf (details in Methods). This vasculature is primarily made up of lignin, cellulose, and hemicellulose, which together are referred to as ‘lignocellulose’18. In order to coat these fibers with Ag, they are first exposed to corona discharge treatment (CDT) using a 10 KV handheld corona surface treater, which generates a partial anionic charge on the fibers. This, when submerged in an Ag microparticle ink incorporating protonated polyethylenimine (PEI) as an adhesion promoter, allows Ag particles to bind strongly to the lignocellulose microstructures as illustrated in Fig. 3a. CDT is specifically chosen here due to its solvent-free and eco-friendly disposition19. The process can additionally be carried out in air without needing special atmospheric conditions while facilitating the express activation of the polymer surface without altering bulk properties. Figure 3a.1 shows the structure of the lignocellulose quasi-fractals. The corona treatment creates activated sites on the lignocellulose surface by deprotonating the hydroxyl (-OH) and carboxyl (-COOH) functional groups, rendering an effective negative charge on the structure as shown in Fig. 3a.2. The fibers when subsequently submerged in Ag ink containing protonated amine groups (Fig. 3a.3) allow the Ag particles to bind to the lignocellulose, resulting in a highly stable and conducting quasi-fractal Ag electrode as shown in Fig. 3a.4.
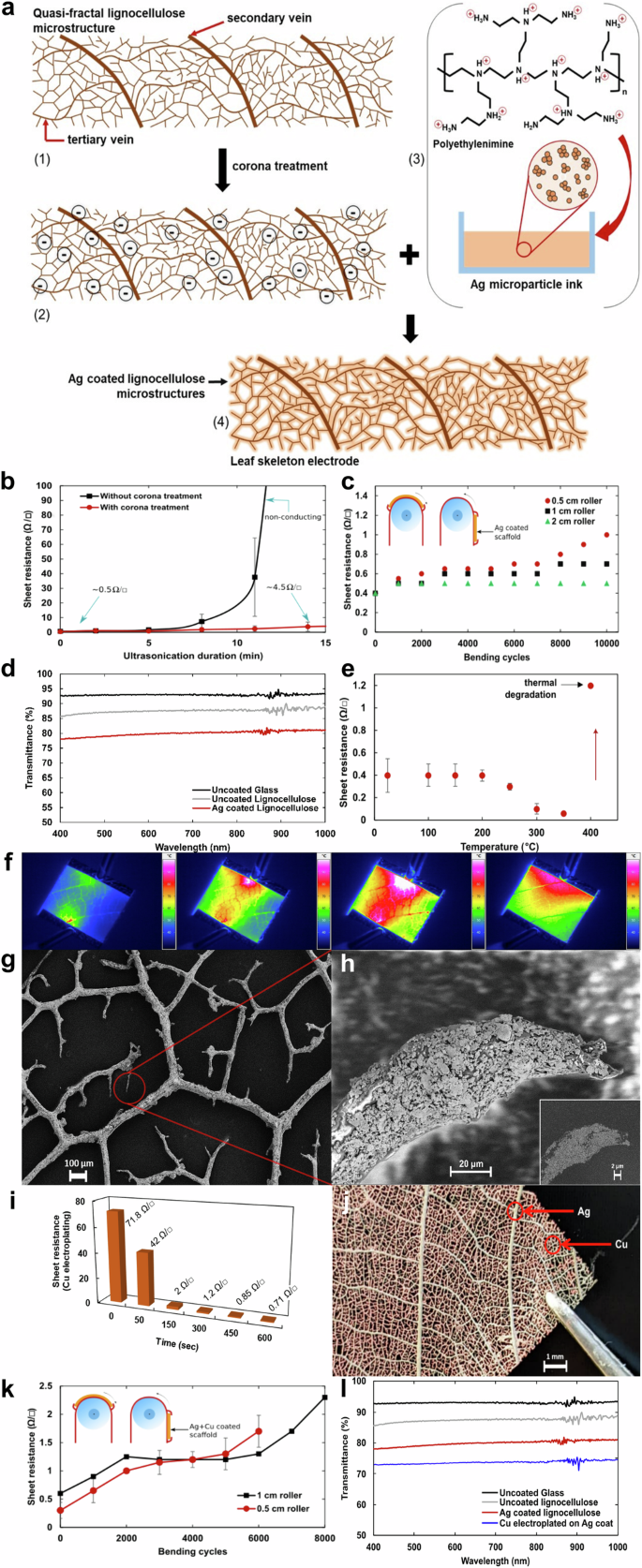
a Illustration of (1) leaf lignocellulose fibers (2) partial negative charge induced onto the lignocellulose fibers after corona treatment (3) protonated PEI in Ag microparticle ink (4) Ag microparticles preferentially adhere to corona treated lignocellulose fibers resulting in a highly conducting, free-standing, quasi-fractal structure. b Change in resistance upon ultrasonic agitation of Ag coated lignocellulose structures with and without corona treatment (c) Change in sheet resistance during 10,000 bend-relax cycles over 5 mm 10 mm and 20 mm rollers (d) Light transmittance measurements of glass, uncoated lignocellulose structures and Ag coated lignocellulose structures (with corona pre-treatment) (e) Change in resistance with respect to temperature (f) Rise in temperature with respect to 4 A of current flow (1) heat distribution 2 s after application of power (2) 4 s after application of power (3) heat spreading across the entire surface of the electrode 6 s after the application of power (4) the quasi-fractal structure allowing for prompt heat dissipation 5 s after removal of applied power (g) SEM image of the Ag coated scaffolds showing their quasi-fractal structure (h) Ag particles thoroughly coating the fibers all the way to the edges (scale bar 100 µm) inset: the distribution of Ag particles (in gray) using energy selective backscattered (EsB) electron detection (scale bar 2 µm) (i) Variation of sheet resistance over the duration of copper electroplating of Ag pre-treated scaffolds (j) Image of the Cu coated lignocellulose scaffold (k) Change in sheet resistance during 8000 (1 cm roller) and 6000 (0.5 cm roller) bend-relax cycles of the Cu electroplated Ag samples (standard deviation of each data point did not exceed ± 0.5 Ω/sq. l Transmittance of the leaf scaffolds after Cu electroplating on Ag for 2 min drops from ~80% to ~75%.
The adhesion of the Ag particles to the lignocellulose scaffolds with and without the corona treatment is compared by ultrasonicating the samples as shown in Fig. 3b. Here, each data point combines the measurements obtained from 5 different 2.5 × 2.5 cm2 electrodes. Samples without the corona treatment begin losing conductivity soon and are completely insulating within 10 min of ultrasonic agitation. However, the corona treated samples retain conduction and the effective resistance only rises by 4Ω even after 18 min in an ultrasonic bath. The data shows that the pre-treatment grants excellent adhesion and thus reliably high conductance to the resulting quasi-fractal electrodes. Additionally, although pristine untreated lignocellulose fibers allow Ag particles to naturally adhere, the results indicate that the adhesion is not stable and fibers loose conductivity easily upon mechanical agitation.
Mechanical stability measurements of the Ag coated lignocellulose structures are shown in Fig. 3c and are measured using a custom-built bending machine. The resistance of 2.5 × 2.5 cm2 samples is measured at regular intervals while they are bent over metal cylinders with diameters of 5 mm, 10 mm and 20 mm to a total of 10,000 bending cycles for each. It is apparent that the Ag coating after corona treatment results in a robust bonding and a negligible change in sheet-resistance is observed even after 10,000 bending cycles over 10 mm and 20 mm rollers while the resistance only increases from 0.4 Ω to 1 Ω after 10,000 bending cycles over a 5 mm diameter roller.
Light transmittance measurements of the Ag coated lignocellulose structures show a maximum transparency value of 80% at 550 nm (Fig. 3d). Although uncoated glass has the highest transmittance values (ranging round 92%), uncoated, pristine Magnolia leaf-skeletons used in this study have transmittances generally over 85% (Fig. 1b). The gap in transmittance of about 6–7% between the uncoated and Ag-coated structures may be further reduced by optimizing the coating technique to achieve improved transmittance values. In previous studies, the transmittance and sheet resistance of ITO-coated glass has been measured to be about 85%20 and around 15 Ω/□21 respectively. With an average sheet resistance of 0.5 Ω/□ and the 80% transmittance of the Ag coated lignocellulose fibers, the figure of merit (FOM) calculation was based on the FOM function developed by Cisneros-Contreras et al. 22, who improved upon Haacke’s original figure of merit function23 for transparent electrodes. The ‘FOM Haacke High Resolution’ (FOMH-HR) is given as:
$$\phi _H-HR=\fracT{\rootn\ofR_\square }$$
(1)
where ‘\(\phi _H-HR\)’ is the FOM in units of Ω-1 and ‘T’ is the transmittance at 550 nm. ‘R□’ is the measured sheet resistance with ‘n’ having a recommended value of 10.
The figure of merit for the quasi-fractal lignocellulose electrodes presented in this work is around 0.85 Ω−1. Considering the literature on transparent electrodes, FOMs greater than 0.45 Ω−1 are already deemed suitable for applications in photovoltaics, light emitting diodes (LEDs), gas sensors, thermal collectors etc.24. Additionally, the FOM reported in this publication is commensurate with and improves upon some of the state-of-the-art results reported previously using more sophisticated approaches24,25,26,27. Leaf based electrodes offer further advantages by being fabricated in ambient conditions without the use of energy intensive techniques such as sputtering or physical vapor deposition (PVD). Finally, the quasi-fractal structure of the electrodes also naturally enhances their electro-optical performance as multiple studies into bio-inspired quasi-fractal electrodes have shown1,10,11,12.
Next, the thermal response of the Ag coated lignocellulose is studied by heating 2.5 × 2.5 cm2 samples in open air while monitoring the change in resistance. It can be seen in Fig. 3e that the sheet resistance remains unchanged until about 200 °C after which a drop in resistance occurs due to the sintering of Ag until 300 °C. Any heating beyond this point results in the substrate loosing structural integrity and fracturing at the slightest contact.
Electro-thermal stability of the lignocellulose-Ag electrodes is measured by passing a direct current (DC) from a constant current source up till a value of 4 A while temperature readings are recorded in parallel. Figure 3f shows that the heat generation from 4 A of current flow stays around 100 °C due to the high conductivity of the electrodes along with the excellent heat dissipation provided by the quasi-fractal structure. The temperature was measured simultaneously with a temperature sensor in thermal contact with the electrodes and a non-contact infrared thermometer. Experiments show that the electrodes can effectively handle currents as high as 6 A indefinitely. Destructive testing yields a value of 6.4 A at which point the electrodes begin emitting tendrils of smoke.
Figure 3g shows the SEM image of the Ag coated scaffolds, and the tip of a random cantilever microfiber is imaged in Fig. 3h. As is evident, the fibers are thoroughly covered with Ag microparticles even at the ends of the fibers and the inset to Fig. 3h shows the extent of metallization using energy selective backscattered (EsB) electron detection (scale bar 2 µm).
Cu electroplating
Although excellent results can be achieved using the aforementioned method of Ag microparticle coating, as shown in Fig. 3a–h, it would be economically beneficial if Ag is replaced with Cu. Direct adhesion of Cu with this method however is not straightforward since Cu in a dispersion needs to be protected from oxidation until the time of coating. This demands either chemically stabilizing Cu or coating the particles with an oxygen barrier which tends to increase the complexity of the process.
Here, we implement a facile method for minimizing the use of Ag and implementing Cu without compromising on the high conductivity and quasi-transparency of the resulting Cu coated electrodes. This is done by first diluting the Ag ink with organic solvent (details in Methods) to reduce its viscosity by a full order of magnitude without changing the original Ag microparticle content. This results in a proportionally low conductivity coating when the CDT treated scaffolds are metallized (~2 mg Ag/ 2.5 cm2 scaffold). The sheet resistance increases by about two orders of magnitude (~70 Ω/□) compared to what is achieved with the undiluted ink (0.5 Ω/□) ( ~7 mg Ag/ 2.5 cm2 scaffold), as can be seen at the zero-second mark in Fig. 3i. This minimal amount of Ag acts as a seed layer for the deposition of Cu during electroplating in a CuSO4.5H2O bath. This facile process results in a Cu based metallization of lignocellulose scaffolds and the electrode resulting from this process is shown in Fig. 3j. Figure 3i also shows the change in sheet resistance over the electrodeposition time. A longer duration of applied power (~200 mW) results in a thicker Cu coating and the results shown in Fig. 3i indicate that a conductivity comparable to that achieved with the purely Ag-based process can be achieved within 5–7 min for a 2.5 × 2.5 cm2 scaffold. The Cu coated scaffold shown in Fig. 3j underwent electroplating for 45 s and hence still shows the underlying Ag seed layer. Figure 3k shows the bending measurements performed on the samples where Cu electroplating was performed on the Ag seed layers. The results show that the sheet resistance increases to twice the values shown in Fig. 3c (for purely Ag coated scaffolds) after 6000 bending cycles on 1 cm and 0.5 cm rollers. This indicates that, although electroplating is effective, the adhesion of the electroplated Cu requires optimization for higher mechanical reliability. This improvement in adhesion of electroplated Cu on Ag may be achieved based on previously reported techniques28,29. Figure 3l shows that the deposition of a Cu layer over the Ag coat reduces the transparency of the conducting scaffold by about 5%.
Effect on pathogens
The Oligodynamic Effect refers to the phenomenon30 that some metals display highly toxic effects on microbial cells. Metals most notably examined for their oligodynamic properties are generally heavy metals like copper31, gold32, silver33, zinc34, and their alloys (for example with magnesium)35. The toxic effect of metal ions on cells36 have been correlated with multiple mechanisms such as protein denaturation, binding to mitochondrial or nuclear DNA and limiting cell multiplication37,38, or directly causing cell membranes to fatally rupture39,40. Most of these mechanisms have been extremely effective against viruses41 (including the SARS-CoV-2 virus42), bacteria43, fungi44, moulds45, and spores46.
Escherichia coli (E. coli) is a bacterial strain belonging to fecal coliforms, typically present in contaminated water and food, and frequently responsible for food poisoning in humans. Moreover, it also causes severe instances of urinary tract infections and gastroenteritis among humans. In turn, fecal bacteria belong to a wider group of total coliforms that are found in soil and water along with human and animal waste.
The setup used to quantify the efficacy of the metallized lignocellulose scaffolds against microbiologically contaminated water comprised stacks of these placed into containers holding contaminated water whilst under agitation for 30 min (Fig. 4a). Various parameters, such as the number of stacks (each stack contained 10 scaffolds measuring 6 cm in diameter), the type of bacterial strains, and the voltage applied across the scaffolds, were varied in this setup to study the oligodynamic efficacy.
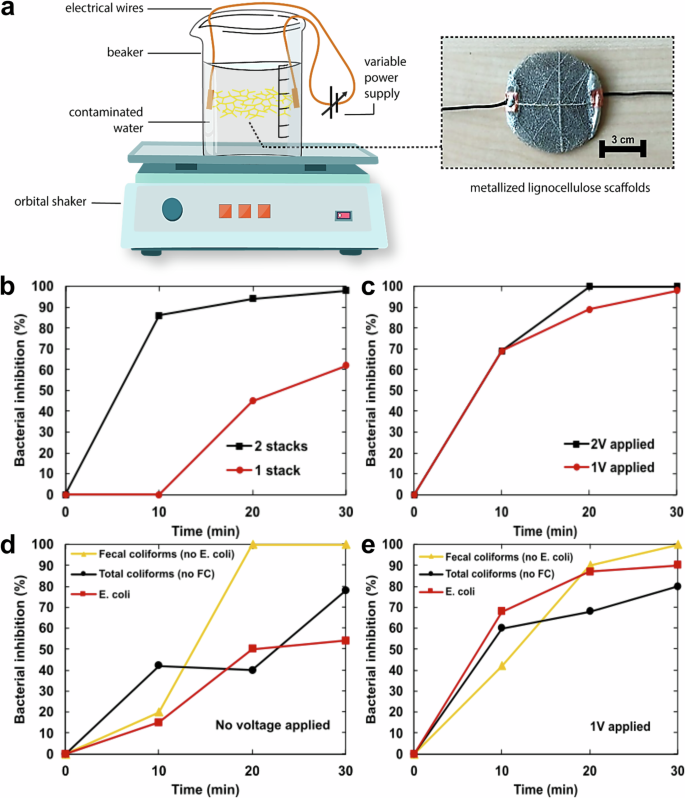
a Basic illustration of the measurement setup used to measure the oligodynamic effect on contaminated water. b E. coli bacterium elimination over a period of 30 min with no voltage applied to the metallized scaffolds (only one stack of metallized scaffolds was used for these experiments) (c) upon the application of 1 Volt (red) and 2 Volts (black) (d) Inhibition of fecal coliforms (FC), total coliforms (TC) and E. coli without any applied voltage (e) Inhibition of fecal coliforms (FC), total coliforms (TC) and E. coli with 1 V applied to the metallized lignocellulose scaffolds. The results represent the average values from at least two independent experiments and each experiment was repeated twice.
The number of Ag coated lignocellulose scaffolds played an important role in the reduction of E. coli concentration during the 30-min duration. As shown in Fig. 4b, the bacterial reduction appears directly linked with the number of Ag coated scaffolds, since where a single stack of scaffolds exhibited an almost 50% inhibition of bacterial growth, two stacks reached an almost 100% inhibition after just 20 min of exposure. This is attributed to the increased concentration of Ag cations released into the water in proportion to the increase in Ag coated stacks and has been previously reported to be a significant contributor to the oligodynamic efficacy of metals like Ag14.
However, the inhibition of E. coli bacterial strains using a single stack of metallized lignocellulose scaffolds reached almost 100% inhibition upon the application of 1 V DC across the stack as shown in Fig. 4c. Basically, the antibacterial effect of a single stack of scaffolds was doubled when just 1 V was applied across it and the efficacy of E. coli inhibition was close to 100% after 30 min of applying 1 V and 20 min of applying 2 V to the metallized lignocellulose electrodes, demonstrating no significant differences in bacterial inhibition (Fig. 4c). These results indicate that the oligodynamic effect, when bolstered with a current flow, can eliminate gram- negative bacterial species such as E. coli, known to be resistant against not only the standard oligodynamic effect (i.e., without the applied voltage as seen in Fig. 4b) but also against many antibiotics, soaps, detergents, and natural enzymes involved in the immune response47,48,49.
Among the bacterial community fecal coliforms serve a crucial role in indicating whether aquatic systems are polluted or not. Additionally, they are utilized in the biodegradation of a diverse array of pollutants within wastewater in order to mitigate their impact on the environment. However, despite efforts, the treatment of industrial and domestic wastewater is not entirely efficient, leading to the release of pollutants and bacterial loads into the environment. It is therefore imperative to reduce the bacterial load originating from Wastewater Treatment Plants (WWTPs) to prevent environmental contamination. Hence, the efficacy and specificity of Ag-coated lignocellulose scaffolds were assessed next against real municipal wastewater samples.
The process involved first quantifying the presence of fecal coliforms (FC), total coliforms (TC) and E. coli present in the municipal wastewater samples before implementing a single stack of metallized lignocellulose scaffolds. The bacterial attrition was subsequently measured in the absence (Fig. 4d) and presence (Fig. 4e) of an applied voltage of 1 V. In the absence of an externally applied voltage, a single stack of scaffolds could eliminate fecal coliforms with 100% efficiency within 20 min. However, the same setup simultaneously had almost no effect on the Total coliforms in 30 min. E. coli inhibition was also low with this setup and only about 50% inhibition could be achieved in 30 min (Fig. 4d). However, when a potential difference of 1 V was applied across the scaffolds, a drastic and consistent increase in pathogenic attrition rates was observed for E.coli and Total coliforms (as shown in Fig. 4e) showing an 80% inhibition of TC and about 90% inhibition of E. coli. As expected, the fecal coliform inhibition also remained high as before.
Measurements performed using Inductively Coupled Plasma Mass Spectrometry (ICP-MS) showed that 2.15 µg of Ag+ cations were released into 100 ml of water in 30 min irrespective of whether an external voltage was applied or not. However, the improvement in bacterial inhibition upon the application of a voltage may be caused by the electrons injected into the quasi-fractal surface of the metallized scaffolds creating oxidation sites that interfere with ion flux across bacterial cell membranes. The very rough surface of the electrodes (Fig. 3g, h) on a sub 100 µm scale may also result in an extremely high electric field density on such sharp corners, leading to a direct electrostatic breakdown of the cell membranes. Further research is required to ascertain the precise mechanisms contributing to the high efficacy of this system under an applied voltage. The results however indicate that such economically metallized micro-fractal structures have the potential to offer a sustainable means to manufacture bio-derived sieves that can decontaminate water passing through them. The high efficacy of the approach also provides sustainable alternatives for wastewater treatment along with maintaining the potabilitiy of drinking water over long periods of time, especially in famine-stricken or arid locations of the planet.
Therefore, leaf-derived quasi-fractal lignocellulose scaffolds can be metallized using a novel Ag microparticle-based methodology that eschews the use of excessive chemical treatments or nanoparticles. The properties of the resulting self-standing, flexible, quasi-transparent electrodes are found to be at par with the state-of-the-art for transparent electrodes used in thin film electronics with the demonstration of sheet resistances below 1 Ω/□ and light transmittance of 80%. It is shown that not only is it a new method to fabricate reliable leaf-based conducting quasi-fractals, but it can also be used to purify water contaminated with bacterial pathogens like E. coli and fecal coliforms. We demonstrated a high, naturally existing propensity of Ag coated lignocellulose scaffolds towards eliminating fecal coliforms with up to 100% efficacy. We also show that this antibacterial effect can be significantly widened to cover bacterial groups such as E. coli and other groups belonging to total coliforms by the application of a small electrical potential (1 V) to the Ag coated lignocellulose scaffolds. The low-cost fabrication and ease of processing makes natural lignocellulose quasi-fractals present in leaves an excellent base for further applications in the field of sustainable flexible electronics as well as water treatment.
link





More Stories
Researchers Reveal Molecular Secrets of Flexible Electronics
Growth Outweighs Uncertainty for Flexible and Printed Electronics: OE-A Survey
SEMI FlexTech Announces 2026 FLEXI Award Winners